Triastek, a Chinese language developer of prescribed drugs produced with additive manufacturing (AM), introduced that the corporate has accomplished the primary in human (FIH) examine of T21, a medicine for ulcerative colitis (UC). As with all of its merchandise, Triastek prints T21 with its proprietary Soften Extrusion Deposition (MED) platform.
To this point, Triastek has positioned a selected emphasis on designing medicine with geometries that may solely be achieved by way of AM, primarily in an effort to management the place a given medicine is launched as soon as ingested, and the speed of launch of the medicine’s energetic substances within the affected person’s system. Relating to T21, these options might result in a breakthrough for people affected by UC, as it’s troublesome for the oral medicines presently available on the market to focus on the colon, particularly.
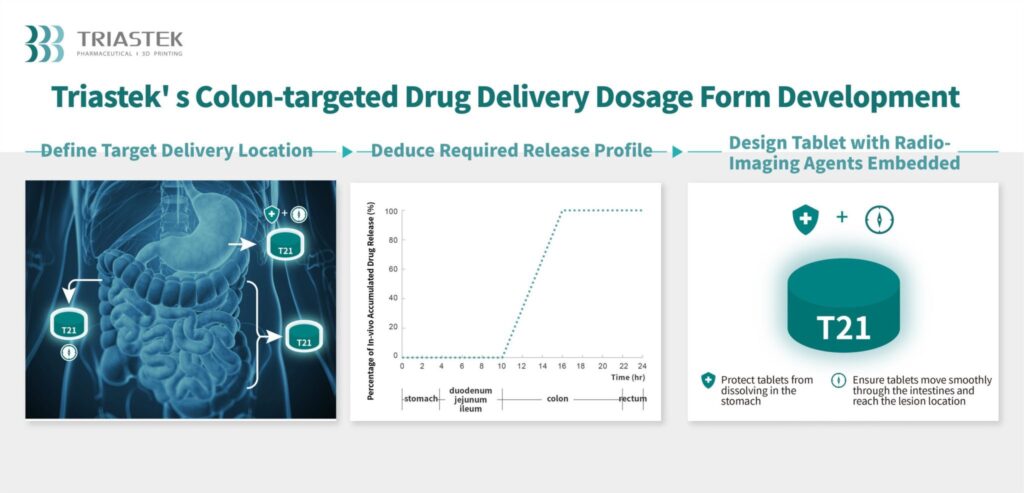
Primarily based on imaging outcomes from the FIH examine, Triastek has proven that upon consumption, T21 tablets are delivered and launched on to the colon. Along with a breakthrough for UC sufferers, this may be a big accelerator for Triastek’s efforts to achieve approval for a New Drug Software (NDA) for T21.

In a press launch about Triastek’s completion of its FIH examine for T21, Professor Xiaoling Li, the co-founder and chief scientific officer of Triastek, defined, “The [FIH] examine information with T21 verifies the exact colon supply functionality of the MED course of, and the platform is poised to turn out to be the novel drug supply system of selection for colon focused new product with both native efficacy or systemic absorption. We hope to proceed showcasing how Triastek’s 3D printing processes can carry technical options to pharmaceutical firms for environment friendly product growth of optimized drug supply, in the end resulting in the flexibility to supply sufferers with extra clinically priceless medicines.”
As I used to be informed by Triastek’s CEO, Dr. Senping Cheng, “…[T21 can mitigate] potential negative effects from systemic publicity [that could result] if the medication was launched elsewhere within the physique.” Once more, because of this utilizing AM for prescribed drugs couldn’t solely result in all of the potential provide chain benefits which have led firms in nearly each trade to start out exploring AM: for prescribed drugs particularly, AM might result in higher high quality merchandise proper now.
After all, that is simpler mentioned than accomplished, with the regulatory course of for medicines happening on a timescale that makes the aerospace sector appear to be a breeze. Then again, the availability shortages within the pharmaceutical trade have been extra intractable than nearly another trade in the previous couple of years, so this might turn out to be a catalyst for an accelerated push to make use of new manufacturing strategies together with AM. This isn’t to recommend that pharmaceutical laws ought to be loosened, however relatively that firms and public businesses ought to work collectively to prioritize the usage of AM and different automation applied sciences within the instances the place constructive outcomes have been confirmed.
Alongside these strains, it’s fascinating that in 2022, world pharma big Eli Lilly introduced that it might collaborate with Triastek to analysis potential new purposes for the MED platform. That is related right here as a result of in April, 2023, the FDA rejected Eli Lilly’s NDA for a brand new UC drug, mirikuzmab. Notably, the rationale for the rejection was associated to issues the FDA had with Eli Lilly’s manufacturing operations in Branchburg, NJ, involving insufficient high quality management.
Pictures courtesy of Triastek
Subscribe to Our E mail Publication
Keep up-to-date on all the newest information from the 3D printing trade and obtain data and presents from third get together distributors.

